Contributed by Gina Brown-Guedira and Suhkwinder Singh
A leaf rust resistance gene designated Lr39 was transferred from Aegilops tauschii accession TA 1675 to the wheat cultivar Wichita and released as the wheat germplasm KS86WGRC02 (2). The gene had been located on the short arm of chromosome 2D by telocentric analysis. However, Cox et al. (1) reported that the gene in KS86WGRC02 did not segregate in crosses with lines havingLr21 and the gene designation was listed as under review. The presence of Lr21 in the line KS86WGRC02 that was distributed as germplasm was also noted by other researchers (McIntosh, personal communication).
Molecular mapping in the cross KS86WGRC02 x Wichita located the leaf rust resistance gene in KS86WGRC02 to the Lr21 locus on chromosome 1DS (4). It was noted that amplification of genomic DNA of Ae. tauschii accession TA 1675 with the STS primer pair KSUD14 that corresponds to a portion of the cloned Lr21 gene did not produce an 885 bp fragment diagnostic for Lr21. However, this fragment is amplified in KS86WGRC02 indicating that the source of the Lr21 gene in germplasm line KS86WGRC02 is not TA 1675. Molecular marker analysis was then done on the original populations from crosses of Wichita with a line having resistance transferred from TA 1675 that was designated as TA 4186 (pedigree = TA 1675/Wichita*3). Our mapping results placed the resistance gene in this line on the distal part of chromosome 2DS, confirming earlier results. The gene is designated Lr39 (3). The most closely linked marker to Lr39 in this population is Xgwm210 that was placed 10.7cM proximal of Lr39. No polymorphic markers in the population are located distal to Lr39.
INFECTION TYPE: When evaluating the effectiveness of resistance conferred by Lr39 in relation to other genes transferred from Ae. tauschii, we noted a striking similarity in infection types of seedlings of TA 4186, the lines U3704 and U4619, and the wheat germplasm KS89WGRC10. U3704 and U4619 are unreleased germplasm lines that have leaf rust resistance genes transferred fromAe. tauschii that we mapped on chromosome 2DS and that did not segregate in crosses with TA 4186. When inoculated with diverse isolates of Puccinia triticina a low infection type (0 to ;) was observed with most isolates on all lines. However, a high infection type (3-4) was observed on the lines after inoculation with the leaf rust race PNMQ.

MAPPING: The germplasm KS89WGRC10 was reported to have a leaf rust resistance gene Lr41 located on the chromosome 1D (Cox et al. 1994). However, we have now mapped the leaf rust resistance gene in KSWGRC10 to the short arm of chromosome 2D. The microsatellite marker Xgdm35 was found to be 0.9 cM proximal to the leaf rust resistance gene in a population of 169 F3 lines from the cross KS89WGRC10 x TAM 107. The Xgdm35 marker was mapped physically on chromosome 2DS using Chinese Spring wheat aneuploid stocks. A fragment of approximately 185bp is amplified in the resistant parent KS89WGRC10 whereas a 280bp fragment is amplified in TAM 107.
ALLELISM: No segregation was observed in F2 populations from the crosses TA 4186 x WX93D246R-1 (300 plants) and KS89WGRC10 x WX93D246R-1 (180 plants). The line WX93D246R-1 is a hexaploid germplasm with leaf rust resistance derived from an accession of Ae. cylindrica. The resistance gene in WX93D246R-1 was mapped on 2DS and has the same race specificity as Lr39(Fritz, unpublished data).
SUMMARY: The genetic mapping of leaf rust resistance in KS89WGRC10 on chromosome 2DS, along with similar race specificity observed for Lr39 and Lr41 suggests that these two genes are the same. This is also supported by results of our allelism studies with line WX93D246R-1 that also has Lr39. To date, Lr39 has been transferred to wheat from at least four accessions of Ae. tauschii of diverse geographic origin as well as an accession of Ae. cylindrica. The closely linked wheat microsatellite marker Xgdm35 should be useful for marker-assisted selection for Lr39 (Lr41).
Markers for Lr39
GDM35 primer sequences and amplification conditions are as in Ref. 5.
Primers sequences:
GDM35-L 5'- CCT GCT CTG CCC TAG ATA CG -3'
GDM35-R 5'- ATG TGA ATG TGA TGC ATG CA -3'
Final concentrations of the reagents used in the PCR amplification (volume: 25 µl)
- 50 ng genomic DNA
- 12.5 pmol each primer
- 0.2 mM each dNTP
- 0.625 U Taq
- 1x PCR buffer
- 2 mM MgCl2
PCR cycling:
- 4 min at 94°C
- 30 cycles of 30s at 94°C, 30s at 55°C, 30s at 72°C
- Final extension for 5 min at 72°C.
PCR products are resolved on 2.3% SFR high resolution agarose gels.
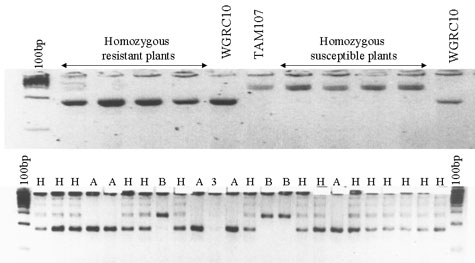
Figure 2. Amplification of wheat microsatellite marker GDM35 in a population of F3 lines from the cross KS89WGRC10 x TAM 107. A= homozygous resistant lines, B = homozygous susceptible lines and H =segregating.
References
1. Leaf rust-resistance genes Lr41, Lr42 and Lr43 transferred from Triticum tauschii to common wheat. Cox, T.S.; Raupp W.J.; Gill, B.S. In: Crop Science, 1994, 34(2):339-343.
2. Registration of KS86WGRC02 Leaf Rust Resistant Hard Red Winter Wheat Germplasm. Gill, B.S.; Raupp, W.J.; Browder, L.E.; Cox, T.S. In:Crop Science, 1988, 28(1):207.
3. Cytogenetic and molecular mapping of the leaf rust resistance gene Lr39 in wheat. Raupp, W.J.; Sukhwinder-Singh; Brown-Guedira, G.L.; Gill, B.S. In: Theoretical and Applied Genetics, 2001, 102(2-3):347-352.
4. An RGA-like marker detects all known Lr21 leaf rust resistance gene family members in Aegilops tauschii and wheat. Huang, L; Gill, B.S. In: Theoretical and Applied Genetics, 2001, 103(6-7):1007-1013.
5. Isolation and mapping of microsatellite markers specific for the D genome of bread wheat. Pestsova, E.; Ganal, M.W.; Röder, M.S. In: Genome, 2000, 43(4)689-697.
